Proust proposed that a compound is always composed of the same proportions of elements by mass.The law of definite composition was proposed by Joseph Proust based on his observations on the composition of chemical compounds.If the sums of the solid or liquid reactants and products are known, any remaining mass can be assigned to gas. An additional useful application of this law is the determination of the masses of gaseous reactants and products. This assumption allows us to represent a chemical reaction as a balanced equation, in which the number of moles of any element involved is the same on both sides of the equation. We can therefore visualize chemical reactions as the rearrangement of atoms and bonds, while the number of atoms involved in a reaction remains unchanged. However, the law of conservation of mass remains a useful concept in chemistry, since the energy produced or consumed in a typical chemical reaction accounts for a minute amount of mass. This amendment incorporates the fact that mass and energy can be converted from one to another. This law was later amended by Einstein in the law of conservation of mass-energy, which describes the fact that the total mass and energy in a system remain constant. In other words, in a chemical reaction, the mass of the products will always be equal to the mass of the reactants. This law states that, despite chemical reactions or physical transformations, mass is conserved-that is, it cannot be created or destroyed-within an isolated system. However, Antoine Lavoisier described the law of conservation of mass (or the principle of mass/matter conservation) as a fundamental principle of physics in 1789. The ancient Greeks first proposed the idea that the total amount of matter in the universe is constant. History of the Law of the Conservation of Mass Antoine Lavoisier: A portrait of Antoine Lavoisier, the scientist credited with the discovery of the law of conservation of mass. Also, a molecule before it undergoes a chemical change. reactant: Any of the participants present at the start of a chemical reaction.product: A chemical substance formed as a result of a chemical reaction.law of conservation of mass: A law that states that mass cannot be created or destroyed it is merely rearranged.The law of conservation of mass is useful for a number of calculations and can be used to solve for unknown masses, such the amount of gas consumed or produced during a reaction.According to the law of conservation of mass, the mass of the products in a chemical reaction must equal the mass of the reactants. 
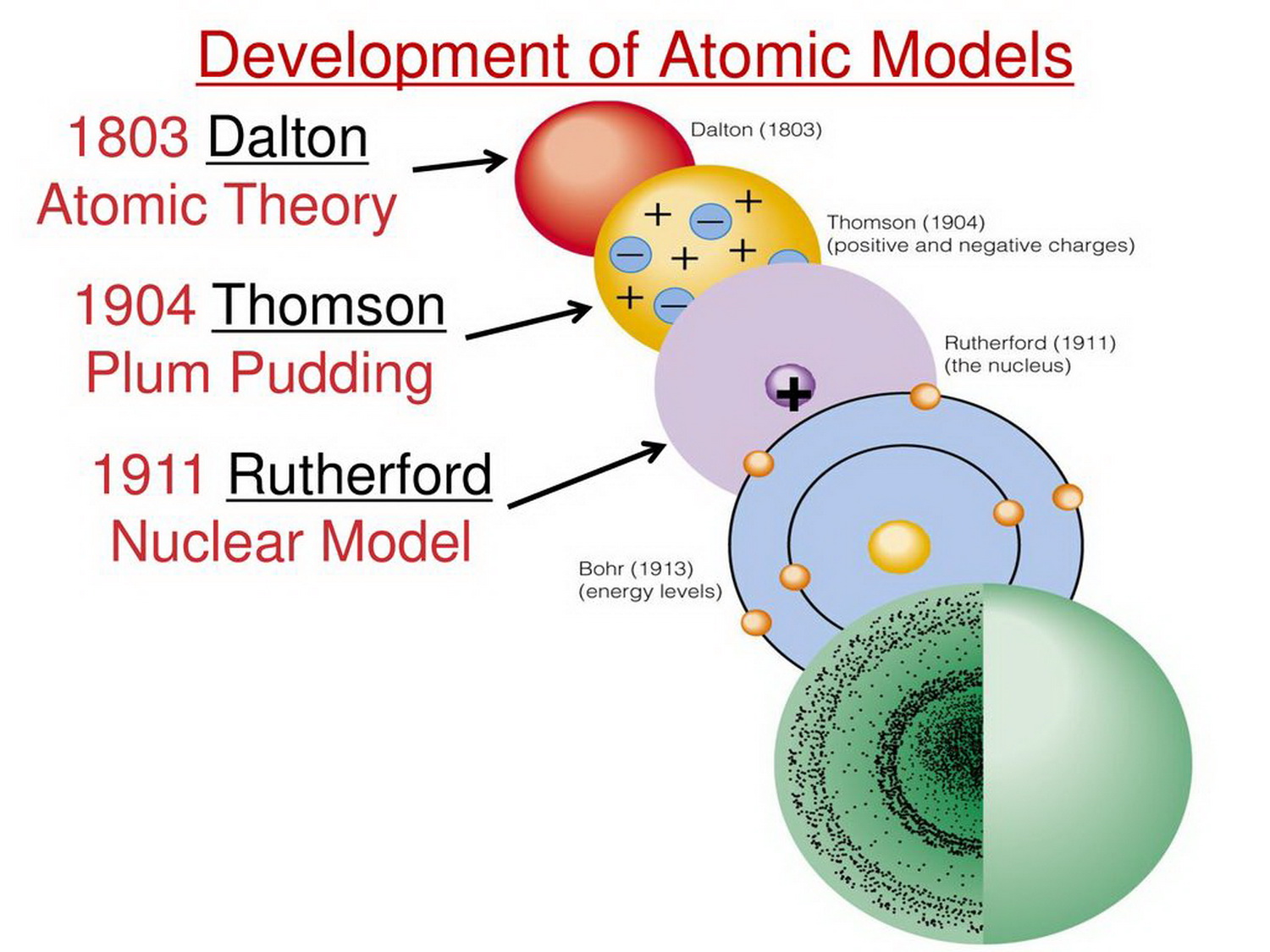
The law of conservation of mass states that mass in an isolated system is neither created nor destroyed by chemical reactions or physical transformations.However, the English chemist and meteorologist John Dalton is credited with the first modern atomic theory, as explained in his A New System of Chemical Philosophy. In 1661, Boyle presented a discussion of atoms in his The Sceptical Chymist. The concept of the atom was revisited and elaborated upon by many scientists and philosophers, including Galileo, Newton, Boyle, and Lavoisier. However, these ideas were largely ignored at the time, as most philosophers favored the Aristotelian perspective. Democritus proposed that different types and combinations of these particles were responsible for the various forms of matter. The Greeks called these particles atomos, meaning indivisible, and the modern word “atom” is derived from this term. This idea was recorded as early as the fifth century BCE by Leucippus and Democritus. Matter is composed of indivisible building blocks. law of multiple proportions: The law stating that reactants will always combine in set whole number ratios.atom: The smallest possible amount of matter that still retains its identity as a chemical element, now known to consist of a nucleus surrounded by electrons.John Dalton, an English chemist and meteorologist, is credited with the first modern atomic theory based on his experiments with atmospheric gases.The idea of an indivisible particle was further elaborated upon and explored by a number of scientists and philosophers, including Galileo, Newton, Boyle, Lavoisier, and Dalton.The ancient Greek philosophers Democritus and Leucippus recorded the concept of the atomos, an indivisible building block of matter, as early as the 5th century BCE. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
